3D
Mol
Similar
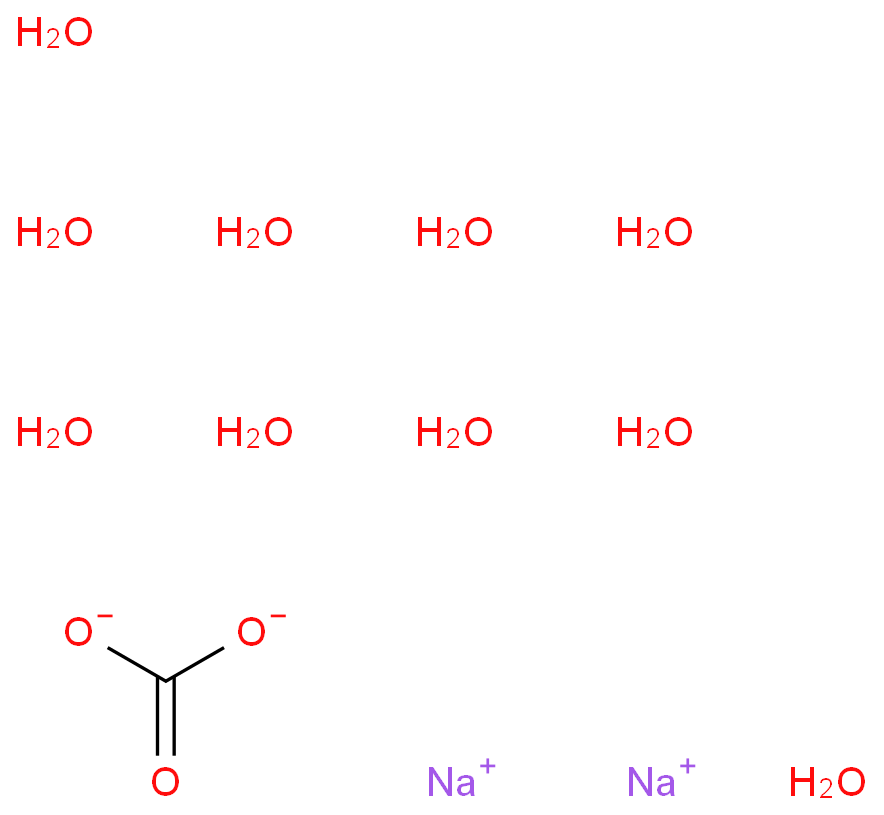
Sodium selenate decahydrate
- Iupac Name:disodium;selenate;decahydrate
- CAS No.: 10102-23-5
- Molecular Weight:369.097
- Modify Date.: 2022-11-25 07:46
- Introduction:
Sodium selenate decahydrate, with the chemical formula Na2SeO4·10H2O and CAS registry number 10102-23-5, is a compound that contains sodium, selenium, oxygen, and water molecules. It is a white crystalline solid that is highly soluble in water. Sodium selenate decahydrate is commonly used as a source of selenium in various industries, including agriculture, pharmaceuticals, and electronics. It is often added to fertilizers to provide plants with essential selenium nutrients. In the pharmaceutical industry, it is used in the production of selenium supplements and medications. Sodium selenate decahydrate also finds applications in the manufacturing of glass, ceramics, and pigments. It is important to handle this compound with care, as it can be toxic if ingested or inhaled in large quantities. Overall, sodium selenate decahydrate plays a crucial role in various industries and contributes to the advancement of technology and human well-being.
View more+
Request For Quotation
1. Names and Identifiers
- 1.1 Name
- Sodium selenate decahydrate
- 1.2 Synonyms
disodium,selenate,decahydrate EINECS 236-501-8 MFCD00149174 selenicacid,disodiumsalt,decahydrate SODIUM SELENATE 10H2O Sodium Selenite Decahydrate SodiumSelenateGr
- 1.3 CAS No.
- 10102-23-5
- 1.4 CID
- 202320
- 1.5 EINECS(EC#)
- 236-501-8
- 1.6 Molecular Formula
- H20Na2O14Se (isomer)
- 1.7 Inchi
- InChI=1S/2Na.H2O4Se.10H2O/c;;1-5(2,3)4;;;;;;;;;;/h;;(H2,1,2,3,4);10*1H2/q2*+1;;;;;;;;;;;/p-2
- 1.8 InChIkey
- TYVNFQRMDPFYPC-UHFFFAOYSA-L
- 1.9 Canonical Smiles
- O.O.O.O.O.O.O.O.O.O.[O-][Se](=O)(=O)[O-].[Na+].[Na+]
- 1.10 Isomers Smiles
- O.O.O.O.O.O.O.O.O.O.[O-][Se](=O)(=O)[O-].[Na+].[Na+]
2. Properties
- 2.1 Density
- 1.6
- 2.1 Melting point
- 35℃ (dec.)
- 2.1 Boiling point
- °Cat760mmHg
- 2.1 Flash Point
- °C
- 2.1 Precise Quality
- 369.98100
- 2.1 PSA
- 172.56000
- 2.1 logP
- -1.49900
- 2.1 Appearance
- white crystals
- 2.2 Storage
- Ambient temperatures.
- 2.3 Chemical Properties
- white crystal(s) [MER06]
- 2.4 Color/Form
- White crystals /Decahydrate/
- 2.5 Water Solubility
- Soluble in water.
- 2.6 StorageTemp
- Keep in a cool, dry, dark location in a tightly sealed container or cylinder. Keep away from incompatible materials, ignition sources and untrained individuals. Secure and label area. Protect containers/cylinders from physical damage.
3. Use and Manufacturing
- 3.1 GHS Classification
- Signal: Danger
GHS Hazard Statements
Aggregated GHS information provided by 26 companies from 2 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
H301 (100%): Toxic if swallowed [Danger Acute toxicity, oral]
H331 (100%): Toxic if inhaled [Danger Acute toxicity, inhalation]
H373 (100%): Causes damage to organs through prolonged or repeated exposure [Warning Specific target organ toxicity, repeated exposure]
H400 (100%): Very toxic to aquatic life [Warning Hazardous to the aquatic environment, acute hazard]
H410 (100%): Very toxic to aquatic life with long lasting effects [Warning Hazardous to the aquatic environment, long-term hazard]
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Precautionary Statement Codes
P260, P261, P264, P270, P271, P273, P301+P310, P304+P340, P311, P314, P321, P330, P391, P403+P233, P405, and P501
- View all
- 3.2 Usage
- Insecticide in some horticultural applications.
4. Safety and Handling
- 4.1 Symbol
- GHS06, GHS08, GHS09
- 4.1 Hazard Codes
- T; N
- 4.1 Signal Word
- Danger
- 4.1 Risk Statements
- R23/25;R33;R50/53
- 4.1 Safety Statements
- S20/21;S28;S45;S60;S61
- 4.1 Packing Group
- I
- 4.1 Other Preventative Measures
- SRP: The scientific literature for the use of contact lenses in industry is conflicting. The benefit or detrimental effects of wearing contact lenses depend not only upon the substance, but also on factors including the form of the substance, characteristics and duration of the exposure, the uses of other eye protection equipment, and the hygiene of the lenses. However, there may be individual substances whose irritating or corrosive properties are such that the wearing of contact lenses would be harmful to the eye. In those specific cases, contact lenses should not be worn. In any event, the usual eye protection equipment should be worn even when contact lenses are in place.
- 4.2 Hazard Class
- 6.1
- 4.2 Hazard Declaration
- H301-H331-H373-H410
- 4.2 DisposalMethods
- SRP: At the time of review, criteria for land treatment or burial (sanitary landfill) disposal practices are subject to significant revision. Prior to implementing land disposal of waste residue (including waste sludge), consult with environmental regulatory agencies for guidance on acceptable disposal practices.
Recycling: As the price of selenium is very high, recovery of it by processing selenium-containing waste is economical. For example, selenium refineries are equipped with wet scrubbers that employ an approx 50% soln of hydrobromic acid containing free bromine. The acid fumes are adsorbed in a lime and soda bed, and the selenium separated from the hydrobromic acid soln by distillation is recycled. Otherwise, solid or sludgy selenium-containing wastes from the manufacture or repair of xerox drums or from paint production are disposed of in special dumps. Recommendable methods: Chemical reduction, solidification & landfill. Not recommendable methods: Thermal destruction & discharge to sewer. Peer review: Reduction with SO2 /sulfur dioxide/ to SE /selenium/ metal. Do not use metal and acids to reduce disodium selenate as this will produce toxic gaseous hydrogen selenide. (Peer-review conclusions of an IRPTC expert consultation (May 1985)) - View all
- 4.3 RIDADR
- UN 2630
- 4.3 Safety Profile
- Poison by intraperitoneal route. When heated to decomposition it emits toxic fumes of Se Sodium selenate decahydrateSupplier
- 4.4 Caution Statement
- P261-P273-P301 + P310-P311-P501
- 4.4 WGK Germany
- 3
- 4.4 RTECS
- VS6720000
- 4.4 Safety
-
Poison by intraperitoneal route. When heated to decomposition it emits toxic fumes of Se.Safety information of Sodium selenate decahydrate (CAS NO.10102-23-5):
Hazard Codes  T,
T, N
N
Risk Statements
23/25 Toxic by inhalation and if swallowed
33 Danger of cumulative effects
50/53 Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
Safety Statements
20/21 When using, do not eat, drink or smoke
28 After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
45 In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
60 This material and/or its container must be disposed of as hazardous waste
61 Avoid release to the environment. Refer to special instructions safety data sheet
RIDADR UN 2630 6.1/PG 1
WGK Germany 3
RTECS VS6720000
HazardClass 6.1
PackingGroup I
- View all
- 4.5 Toxicity
- LD50 i.p. in mice: 18.45 mg/kg (Nofre)
5. MSDS
2.Hazard identification
2.1 Classification of the substance or mixture
Acute toxicity - Oral, Category 3
Acute toxicity - Inhalation, Category 3
Specific target organ toxicity \u2013 repeated exposure, Category 2
Hazardous to the aquatic environment, short-term (Acute) - Category Acute 1
Hazardous to the aquatic environment, long-term (Chronic) - Category Chronic 1
2.2 GHS label elements, including precautionary statements
| Pictogram(s) |    |
| Signal word | Danger |
| Hazard statement(s) | H301 Toxic if swallowed H331 Toxic if inhaled H373 May cause damage to organs through prolonged or repeated exposure H410 Very toxic to aquatic life with long lasting effects |
| Precautionary statement(s) | |
| Prevention | P264 Wash ... thoroughly after handling. P270 Do not eat, drink or smoke when using this product. P261 Avoid breathing dust/fume/gas/mist/vapours/spray. P271 Use only outdoors or in a well-ventilated area. P260 Do not breathe dust/fume/gas/mist/vapours/spray. P273 Avoid release to the environment. |
| Response | P301+P310 IF SWALLOWED: Immediately call a POISON CENTER/doctor/\u2026 P321 Specific treatment (see ... on this label). P330 Rinse mouth. P304+P340 IF INHALED: Remove person to fresh air and keep comfortable for breathing. P311 Call a POISON CENTER/doctor/\u2026 P314 Get medical advice/attention if you feel unwell. P391 Collect spillage. |
| Storage | P405 Store locked up. P403+P233 Store in a well-ventilated place. Keep container tightly closed. |
| Disposal | P501 Dispose of contents/container to ... |
2.3 Other hazards which do not result in classification
none
View all
6. Other Information
- 6.0 Merck
- 14,8671
- 6.1 Chemical Properties
- white crystal(s) [MER06]
- 6.2 Uses
- Insecticide in some horticultural applications.
- 6.3 Safety Profile
- Poison by intraperitoneal route. When heated to decomposition it emits toxic fumes of Se
- 6.4 Usage
- Sodium selenate decahydrate is used as a feed additive, as a mineral supplement, as analytical reagents and in horticultural applications. Further, it is used in manufacturing of glass to counter the color caused by iron impurities. It is also employed in the prevention of abnormal tissue growth. In addition to this, it is used as a micronutrient which prevents a deficiency of copper, manganese and selenium.
- 6.5 Uses
- Sodium Selenate Decahydrate is used in preparation method of Selenium enriched crop cultivation substrate by using crop straw biochar.
7. Computational chemical data
- Molecular Weight: 369.097g/mol
- Molecular Formula: H20Na2O14Se
- Compound Is Canonicalized: True
- XLogP3-AA: null
- Exact Mass: 369.98137
- Monoisotopic Mass: 369.98137
- Complexity: 62.2
- Rotatable Bond Count: 0
- Hydrogen Bond Donor Count: 10
- Hydrogen Bond Acceptor Count: 14
- Topological Polar Surface Area: 90.3
- Heavy Atom Count: 17
- Defined Atom Stereocenter Count: 0
- Undefined Atom Stereocenter Count: 0
- Defined Bond Stereocenter Count: 0
- Undefined Bond Stereocenter Count: 0
- Isotope Atom Count: 0
- Covalently-Bonded Unit Count: 13
- CACTVS Substructure Key Fingerprint: AAADceAAPDAAAAAAIAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAACAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAAA==
8. Recommended Suppliers
Global22Suppliers View all >>
9. Realated Product Infomation
-
1303-96-4
Sodium tetraborate decahydrate
-
13472-36-1
Sodium pyrophosphate decahydrate
-
7727-73-3
SODIUM SULFATE DECAHYDRATE
-
6132-02-1
Sodium carbonate decahydrate
-
24551-51-7
SODIUM CARBONATE DECAHYDRATE
-
13517-17-4
Sodium chromate decahydrate
-
11139-65-4
decahydrate
Recently Updated :
1825621-12-2
1825499-42-0
1645488-02-3
1825634-23-8
1645502-34-6
1645523-80-3
1645361-78-9
1352564-65-8
1645370-26-8
1645576-94-8
Directory
1. Names and Identifiers
- 1.1 Name
- 1.2 Synonyms
- 1.3 CAS No.
- 1.4 CID
- 1.5 EINECS(EC#)
- 1.6 Molecular Formula
- 1.7 Inchi
- 1.8 InChIkey
- 1.9 Canonical Smiles
- 1.10 Isomers Smiles
2. Properties
-
- 2.1 Density
- 2.1 Melting point
- 2.1 Boiling point
- 2.1 Flash Point
- 2.1 Precise Quality
- 2.1 PSA
- 2.1 logP
- 2.1 Appearance
- 2.2 Storage
- 2.3 Chemical Properties
- 2.4 Color/Form
- 2.5 Water Solubility
- 2.6 StorageTemp
3. Use and Manufacturing
-
- 3.1 GHS Classification
- 3.2 Usage
4. Safety and Handling
-
- 4.1 Symbol
- 4.1 Hazard Codes
- 4.1 Signal Word
- 4.1 Risk Statements
- 4.1 Safety Statements
- 4.1 Packing Group
- 4.1 Other Preventative Measures
- 4.2 Hazard Class
- 4.2 Hazard Declaration
- 4.2 DisposalMethods
- 4.3 RIDADR
- 4.3 Safety Profile
- 4.4 Caution Statement
- 4.4 WGK Germany
- 4.4 RTECS
- 4.4 Safety
- 4.5 Toxicity
5. MSDS
6. Other Information
7. Computational chemical data
8. Recommended Suppliers






 T,
T, N
N